
Consumers often wonder how to identify safe and high-quality organ meat supplements. Quality depends on strict sourcing and manufacturing standards. Manufacturers may use different capsule suppliers, which can cause minor differences in supplement appearance, but these changes do not affect safety or health benefits. Companies must follow rigorous testing and regulatory standards to ensure that organ supplements meet label claims and remain effective. Recent findings show that nutritional supplements can vary in quality, making it important to check sourcing and manufacturing details before choosing a product.
Medical Disclaimer: This information is for educational purposes only. Individuals should consult a healthcare professional before using any supplements.
Key Takeaways
Choose organ meat supplements sourced from grass-fed, pasture-raised cattle without hormones or antibiotics for better nutrition and safety.
Look for freeze-dried and non-defatted processing methods to preserve vitamins, minerals, and protein quality in supplements.
Select products made in GMP-certified and FDA-registered facilities to ensure strict manufacturing and safety standards.
Check for clean label practices that avoid fillers, artificial additives, and use transparent ingredient lists.
Prefer single-source bovine organ powders with third-party testing and Certificates of Analysis (COAs) to confirm purity and label accuracy.
Verify supplements undergo microbiological, heavy metal, pesticide, and nutrient testing by ISO 17025-accredited labs for safety.
Review certifications like GMP, FDA registration, ISO 17025, and USDA Organic to confirm quality and ethical sourcing.
Avoid supplements with fillers, vague sourcing, no COA, or poor reviews to protect your health and investment.
Quality of Organ Meat Supplements
Sourcing Standards
Grass-Fed and Pasture-Raised
Beef organ supplements achieve uncompromising quality when brands prioritize sourcing from grass-fed and pasture-raised cattle. These sourcing standards ensure animals graze freely, receive organic feed, and live in humane conditions.
USDA organic standards require livestock to access outdoors, shelter, exercise, fresh air, and sunlight.
Organic management prohibits genetic engineering, ionizing radiation, and sewage sludge.
Brands such as Heart & Soil, Hirsch Organic, and MK Supplements select cattle raised on regenerative farms, supporting natural resource conservation and biodiversity.
Pasture-raised verification and USDA Organic certification confirm that animals are raised sustainably and ethically, which directly impacts the nutrient density of bovine organ powders.
Producers like Kilcoy Nutrition and Grassfat source organs from healthy, locally raised cattle, ensuring low-stress handling and adherence to strict welfare protocols. These practices result in nutrient-dense supplements with superior bioavailability advantages.
Hormone and Antibiotic-Free
High-quality beef organ supplements must come from cattle raised without synthetic hormones or antibiotics.
Organic certification prohibits antibiotics and synthetic hormones, ensuring clean, safe organ meat supplements.
Brands such as Grassfat and Hirsch Organic guarantee hormone- and antibiotic-free sourcing, verified by USDA-accredited certifying agents.
This commitment to purity supports maximum nutrient retention and reduces the risk of contaminants in bovine organ powders.
Third-party testing and Certificates of Analysis (COA) from independent labs further validate the absence of prohibited substances, reinforcing consumer trust in beef organ complex supplements.
Processing Methods
Freeze-Drying
Freeze-drying stands as the gold standard for preserving the nutritional integrity of organ meat supplements.
Leading manufacturers use rapid freezing at -40°C immediately after harvest, halting microbial activity and oxidation. Sublimation under vacuum at low temperatures avoids heat damage, while gentle rinsing with peroxyacetic acid neutralizes microbes without degrading nutrients.
The process retains heat-sensitive vitamins, minerals, and proteins, resulting in nutrient-dense superfoods with high nutrient density.
Aspect | Freeze Drying | Heat Drying |
|---|---|---|
Mechanism | Sublimation of ice under vacuum at low temperatures | Evaporation using hot air at 45–90°C |
Temperature Range | -20°C (primary) to 30–40°C (secondary) | 45–90°C |
Nutrient Retention: Vitamin C | ≥ 90% preserved | 20–60% preserved |
Nutrient Retention: B-complex Vitamins | Losses under 15% | Significant losses due to heat |
Nutrient Retention: Fat-soluble Vitamins (A, D, E, K) | ≥ 95% preserved | 30–50% preserved, oxidation occurs |
Protein Integrity | Preserved in native form | Partial denaturation due to heat |
Processing Time | 24–72 hours | 6–12 hours |
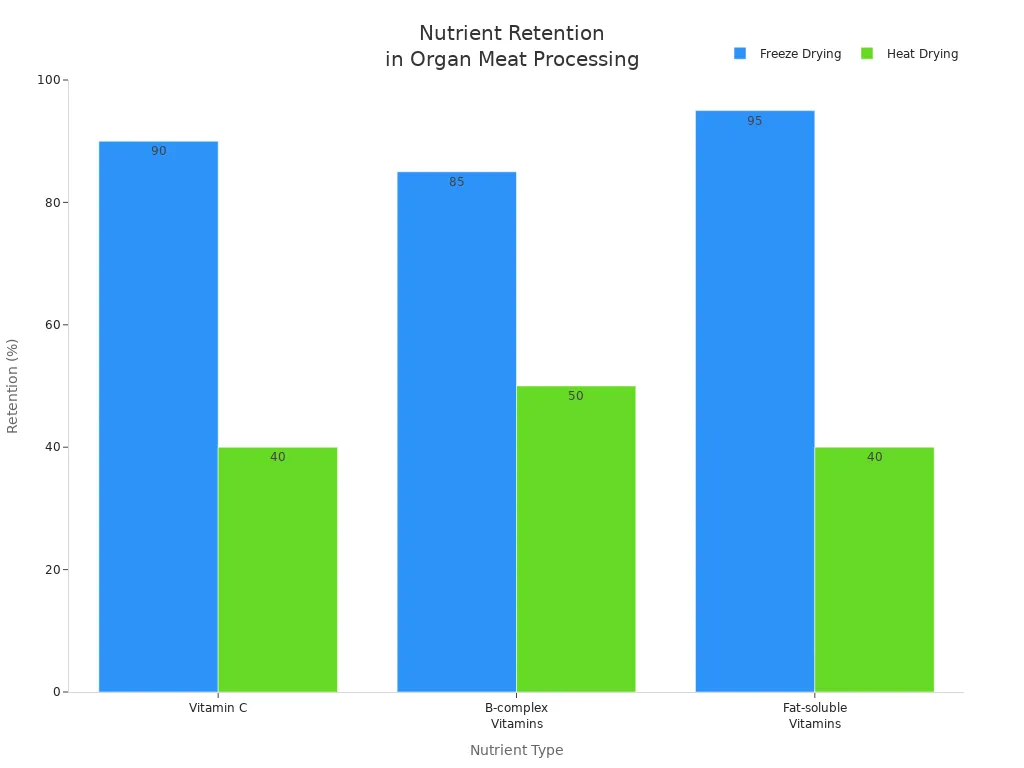
Freeze-dried beef organ supplements retain more vitamins and minerals than those processed with heat. This method ensures maximum nutrient retention and preserves the bioavailability advantages of bovine organ powders.
Non-Defatted Organs
Non-defatted processing keeps organ meats in their natural state, preserving all fats, vitamins, minerals, proteins, and enzymes.
Non-defatted desiccated organs maintain the original ratios found in nature, enhancing nutrient balance and retention.
Brands such as MK Supplements and Heart & Soil use non-defatted organs, resulting in nutrient-dense supplements that deliver the full spectrum of nutrients.
Third-party testing supports purity and safety, confirming that supplements remain free from contaminants.
Freeze-drying and non-defatted processing together produce beef organ supplements with superior retention of nutritional components, supporting optimal health benefits.
Note: Consumers seeking high-quality organ meat supplements should look for products with USDA Organic certification, pasture-raised sourcing, hormone- and antibiotic-free claims, and freeze-dried, non-defatted processing. These standards ensure the highest level of safety, purity, and nutritional value.
Manufacturing Standards for Beef Organ Supplements

GMP Certification
Good Manufacturing Practice (GMP) certification stands as a cornerstone of quality assurance in the production of beef organ supplements. Facilities that achieve GMP certification follow strict protocols for cleanliness, documentation, and process control. These standards help ensure that supplements meet label claims and remain free from contaminants. Advanced Supplements, for example, manufactures beef organ supplements in GMP-certified facilities, demonstrating a commitment to quality control and regulatory compliance. Brands that invest in GMP certification provide consumers with confidence that each batch of supplements undergoes rigorous oversight. GMP-certified manufacturing also supports traceability, allowing companies to track ingredients from sourcing to finished product.
FDA Registration
FDA registration plays a vital role in the safety and traceability of beef organ supplements. Facilities that manufacture, process, pack, or hold food must register with the FDA. This registration allows the FDA to oversee operations and collaborate with state agencies to ensure compliance with safety standards. The following table outlines key FDA requirements and their impact on beef organ supplement manufacturing:
Regulatory Requirement | Description | Impact on Safety and Traceability |
|---|---|---|
Recordkeeping by Renderers | Renderers handling cattle materials must maintain records proving that prohibited materials are excluded from animal feed. These records must be available for FDA inspection. | Ensures traceability of cattle materials and prevents contamination with high-risk materials, supporting safety in beef organ supplement manufacturing. |
Certification and Documentation | Suppliers to renderers must provide certification or documentation confirming segregation procedures to exclude prohibited cattle materials. Renderers must periodically review these certifications. | Enforces supplier accountability and documentation, enhancing traceability and safety by verifying exclusion of prohibited materials. |
Record Retention Period | Records must be kept for at least one year, consistent with existing feed regulations. | Supports traceability by allowing investigation and trace-back of materials within a relevant timeframe. |
Written Procedures for Removal of High-Risk Materials | Rendering firms must have written procedures to remove brain and spinal cord from cattle not inspected and passed for human consumption or verify cattle age if removal is not done. | Prevents inclusion of high-risk materials in feed, directly impacting safety of beef organ supplements. |
FDA Registration Requirement | Facilities manufacturing, processing, packing, or holding food must register with FDA; however, additional registration for firms handling prohibited materials is not currently required. | While registration itself is not a direct safety measure, it facilitates FDA oversight and collaboration with state agencies to ensure compliance, indirectly supporting safety and traceability. |
FDA registration does not certify supplements, but it does enable fda-inspected manufacturing and supports regulatory compliance. This oversight helps protect consumers and ensures that beef organ supplements meet established safety standards.
Clean Label Practices
Clean label practices have become a benchmark for quality in beef organ supplements. Brands that follow clean label principles use transparent ingredient lists and avoid unnecessary additives. The table below highlights the main components of clean label practices:
Component | Description |
|---|---|
Sourcing | Use 100% grass-fed, pasture-raised beef from reputable regions such as New Zealand and Argentina. |
Processing Methods | Prefer freeze-drying to preserve nutrient integrity; avoid heat treatments. |
Formula Purity | Free from fillers, binders, artificial ingredients, hormones, GMOs, and flow agents like magnesium stearate. |
Labeling | Clear and transparent listing of all ingredients. |
Consumers should look for beef organ supplements that display clean label commitments. These practices ensure that supplements deliver pure, nutrient-rich ingredients without hidden additives. Brands that embrace clean label standards often provide detailed sourcing and processing information, supporting both quality and transparency.
Note: Manufacturing transparency and regulatory compliance remain essential for consumer trust. Brands that follow GMP certification, FDA registration, and clean label practices set the highest standards for beef organ supplements.
Ingredient Purity in Bovine Organ Powders
Single-Source vs. Multi-Source
Bovine organ powders can differ greatly based on their source. Single-source powders use organs from cattle raised on one farm or in a specific region. This approach improves traceability and consistency. It also reduces the risk of contamination. Single-source products often come from grass-fed and pasture-raised cattle in countries with strict farming laws, such as New Zealand, Australia, or the USA. These powders usually have higher nutrient density, including vitamins A, B12, CoQ10, selenium, and omega-3 fatty acids. Brands that use single-source organs often conduct third-party testing for heavy metals, pathogens, and pesticides.
Multi-source bovine organ powders blend organs from several unknown sources. This practice can lower transparency and increase safety concerns. Multi-source products may not always undergo the same level of testing. They sometimes include fillers or additives, which can dilute the potency and introduce allergens. Freeze-dried processing remains important for both types, but single-source powders more often use this method exclusively.
Tip: Choose single-source bovine organ powders for better traceability, higher nutrient content, and improved safety.
Avoiding Fillers
Fillers can reduce the effectiveness of bovine organ powders and may cause unwanted reactions. Low-quality supplements often contain rice flour, silicon dioxide, or magnesium stearate. These substances can act as allergens or irritants. They also decrease the concentration of active ingredients.
To detect fillers, consumers should:
Review the supplement facts panel and ingredient list for unnecessary or synthetic ingredients.
Look for products with short ingredient lists that contain only freeze-dried organs.
Check for third-party lab testing and Certificates of Analysis (COAs) to confirm the absence of fillers and contaminants.
High-quality bovine organ powders remain free from harmful additives and deliver pure nutrition.
Label Accuracy
Accurate labeling is essential for consumer trust and safety. Recent studies have found that some beef organ complex supplements do not match their label claims. Noncompliant nutrient content and misleading ingredient lists can pose health risks.
Consumers can identify pure, high-quality bovine organ powders by following these steps:
Check for a simple ingredient list with only bovine-derived ingredients and no additives, artificial colors, or preservatives.
Look for “additive-free” and “non-GMO” labels.
Verify transparency about sourcing, such as grass-fed, pasture-raised cattle and country of origin.
Confirm certifications like ISO 9001/22000, GMP, HACCP, Halal, Kosher, or environmental certifications.
Seek third-party testing and access to lab results or COAs for contaminants.
Prefer brands that share detailed sourcing, processing methods, and batch-specific lab results.
Look for third-party verification seals on packaging.
These steps help ensure that bovine organ powders meet high standards for purity, safety, and nutritional value.
Dietary Supplement Safety and Testing

Microbiological Testing
Microbiological testing plays a critical role in dietary supplement safety, especially for organ meat supplements. Manufacturers follow Good Manufacturing Practice (GMP) and Hazard Analysis and Critical Control Points (HACCP) protocols to control contamination risks. These protocols require routine microbial testing of production environments, equipment, and surfaces. Pathogens such as Listeria monocytogenes and Salmonella can enter during harvesting, washing, or processing. Producers use food-grade peroxyacetic acid rinses to disinfect organs at harvest. They then flash-freeze organs at -40°C to halt microbial activity and preserve nutrients.
The testing process includes several steps:
Initial testing of raw frozen organs for total aerobic microbial count, mold, yeast, and common pathogens like E. coli and Staphylococcus aureus.
Discarding any batches that fail these tests.
Freeze-drying under cold, low-pressure conditions to remove moisture and prevent microbial growth.
Retesting for moisture content, microbial levels, and contaminants after freeze-drying.
Manufacturing in GMP-certified, FDA-inspected facilities with strict sanitation and staff hygiene training.
Using infrared spectroscopy for identity verification and swab testing of capsules to detect contamination during encapsulation and packaging.
Independent third-party laboratory validation of finished products, including microbial counts and mold and yeast screening.
Brands that publish Certificates of Analysis (COA) demonstrate transparency and commitment to dietary supplement safety. These steps help ensure that supplements remain free from harmful microbes and safe for consumption.
Heavy Metal Screening
Heavy metal screening is essential for all supplements, including organ meat supplements. Heavy metals such as lead, cadmium, and mercury can accumulate in animal tissues and pose health risks if present above regulatory limits. International organizations like the Food and Agriculture Organization (FAO), World Health Organization (WHO), and Codex Alimentarius set maximum acceptable limits for heavy metals in meat products. For example, lead should not exceed 0.1 mg/kg, cadmium should remain at or below 0.05 mg/kg, and mercury should not surpass 0.5 mg/kg. The European Union enforces similar standards to protect consumers from toxic effects.
Manufacturers use validated analytical methods, such as high-performance liquid chromatography (HPLC) and other advanced techniques, to detect and quantify heavy metals in supplements. Each production lot undergoes batch testing before release. The production run stays quarantined until lab-tested results confirm safety. Reputable brands also conduct retail blind sample screenings several times a year to verify consistency between tested batches and products sold in stores.
Consumers should look for brands that provide third-party testing for purity and publish heavy metal screening results in their COAs. This practice supports dietary supplement safety and builds trust in the nutritional quality of the product.
Pesticide and Toxin Checks
Pesticide and toxin checks form another pillar of dietary supplement safety. Organ meat supplements may contain residues from environmental exposure or agricultural practices. Manufacturers must establish specifications for contamination limits and use scientifically valid test methods to verify safety. Public quality standards, such as those from the United States Pharmacopeia-National Formulary (USP-NF) and the Dietary Supplements Compendium (DSC), offer validated analytical methods and acceptance criteria for contaminants.
Routine testing includes:
Screening for pesticide residues using advanced analytical techniques.
Checking for mycotoxins and other environmental toxins.
Verifying that all results fall within established safety limits.
Manufacturers often use third-party laboratories accredited to ISO 17025 standards for these tests. Brands like Eurofins USA and Gembra Health exemplify this commitment to rigorous testing. Frequent batch testing and transparent reporting through COAs help ensure that supplements meet safety standards and remain free from harmful substances.
Tip: Always review a supplement’s COA for detailed results on microbiological, heavy metal, and pesticide testing. This step helps confirm that the product is safe, pure, and lab-tested for quality.
Protein and Nutrient Verification
Manufacturers must verify the protein and nutrient content of organ meat supplements to ensure label accuracy and consumer safety. They use a combination of advanced laboratory techniques to analyze the nutritional profile of each batch. Proximate testing quantifies the main components, including protein, fat, fiber, moisture, and ash. This method provides a comprehensive overview of the supplement’s composition.
Amino acid profiling plays a crucial role in determining protein quality. Essential amino acids, which the body cannot produce, must be present in adequate amounts. Laboratories use specialized quantification methods to measure these amino acids and confirm that the supplements deliver complete protein benefits.
Vitamin analysis requires high-precision instruments. Techniques such as high-performance liquid chromatography (HPLC), ultra-performance liquid chromatography (UPLC), gas chromatography (GC), and microbial or fluorometric assays help identify and quantify both fat- and water-soluble vitamins. These methods ensure that the supplements provide the expected levels of vitamins A, B12, D, and others.
Enzyme activity assays, following standards from organizations like AOAC, ISO, and USP, measure the functional activity of enzymes in the supplements. These tests confirm that enzymes remain active after processing and storage, supporting the bioactive potential of the product.
The following table summarizes the main testing methods used for nutrient verification in organ meat supplements:
Nutrient Type | Testing Method(s) | Description |
|---|---|---|
Protein | Proximate testing, Amino acid profiling | Quantifies total protein and assesses quality through essential amino acid analysis. |
Amino Acids | Amino acid quantification methods | Measures individual amino acids to ensure a complete protein profile. |
Vitamins | HPLC, UPLC, GC, Microbial, Fluorometric methods | Identifies and quantifies fat- and water-soluble vitamins. |
Enzymes | Enzyme activity assays (USP, ISO, AOAC methods) | Verifies the presence and activity of functional enzymes. |
Fats, Oils, Fiber, Ash | Proximate analysis methods | Assesses other key nutrients for a complete nutritional profile. |
Manufacturers follow accredited standards to guarantee accuracy and regulatory compliance. These rigorous testing protocols help ensure that supplements deliver the nutrients promised on their labels.
Third-Party and ISO 17025 Lab Testing
Independent laboratory testing stands as a critical safeguard for dietary supplement safety. Leading brands partner with third-party labs accredited to ISO 17025 standards, such as Eurofins USA and Gembra Health. These labs possess the expertise and equipment necessary to conduct precise, unbiased analyses of organ meat supplements.
ISO 17025 accreditation signals that a laboratory meets international standards for technical competence and quality management. This accreditation covers all aspects of testing, from sample handling to data reporting. Brands that use ISO 17025-accredited labs demonstrate a commitment to transparency and consumer trust.
Third-party testing covers a wide range of safety and quality checks, including:
Microbiological screening for pathogens and spoilage organisms.
Heavy metal analysis for contaminants like lead, cadmium, and mercury.
Pesticide and toxin residue testing.
Protein, vitamin, and amino acid verification.
Testing against prohibited substances lists for athletes, ensuring supplements are safe for competitive use.
Manufacturers often test every batch or conduct frequent random sampling. They publish Certificates of Analysis (COA) for each lot, allowing consumers to review detailed results. The COA provides proof that the supplements meet safety and quality standards.
Tip: Always request and review the COA before purchasing organ meat supplements. This document confirms that the product has passed independent, accredited laboratory testing.
Regular third-party and ISO 17025 lab testing helps maintain high standards in the supplement industry. It protects consumers from mislabeled or contaminated products and supports informed decision-making.
Verifying Organ Supplements as a Consumer
Certifications and Accreditations
Consumers can build confidence in bovine organ powders by checking for recognized certifications and accreditations. Reliable supplements often display GMP (Good Manufacturing Practice), FDA registration, ISO 17025 laboratory accreditation, and organic certification. These standards confirm that manufacturers follow strict protocols for safety, sourcing, and quality.
To verify certifications, consumers should:
Review product packaging and official websites for certification logos.
Search for documentation or certification numbers linked to GMP, FDA, ISO, or USDA Organic.
Confirm that third-party organizations, such as US Pharmacopeia (USP), NSF International, UL Solutions, BSCG, or Informed Choice, have tested the supplements.
Check for sourcing details, such as grass-fed or pasture-raised claims, which often accompany organic certification.
A table can help summarize key certifications:
Certification | What It Means | Where to Find It |
|---|---|---|
GMP | Manufacturing follows strict standards | Product label, website |
FDA Registration | Facility registered with FDA | Website, documentation |
ISO 17025 | Lab meets global testing standards | COA, lab reports |
USDA Organic | Sourcing meets organic requirements | Label, certification docs |
These steps help consumers select supplements that meet high standards for safety and purity.
Accessing COAs
Certificates of Analysis (COAs) provide objective proof of supplement safety and ingredient accuracy. Reputable brands offer COAs on product pages or upon request. For example, some companies link third-party COAs directly on their websites, demonstrating transparency and commitment to quality.
Consumers should:
Request a COA from the manufacturer if it is not publicly available.
Review the COA for confirmation of ingredient content, absence of contaminants, and compliance with safety standards.
Check that the COA comes from an independent, ISO 17025-accredited laboratory.
Look for verification of claims such as lab-tested, cruelty-free, GMO-free, and CGMP certification.
A refusal to provide a COA may signal a lack of transparency or possible issues with product quality. Consumers should prioritize supplements with accessible, third-party COAs to ensure consumer confidence.
Tip: When interpreting a COA, verify that the supplement contains the stated active ingredients and meets safety limits for heavy metals, microbes, and pesticides. Third-party certifications from organizations like USP or NSF further confirm purity and potency.
Brand Reputation
Brand reputation plays a vital role in choosing safe bovine organ powders. Reliable companies demonstrate rigorous third-party testing for purity, potency, and safety. They source organs ethically, often from grass-fed cattle in regions known for strict farming standards.
Key indicators of a reputable brand include:
Commitment to sustainability and transparent supply chains.
Clear information about product contents and nutritional benefits.
Emphasis on safety, with contamination-free supplements.
Positive customer feedback and reviews on independent platforms.
Responsive customer service that answers questions about sourcing, testing, and certifications.
Consumers should review feedback from other buyers and communicate directly with brands to clarify any concerns. Companies that value transparency and quality will provide detailed answers and documentation.
Medical Disclaimer: The information provided in this article is for informational purposes only. It does not constitute medical advice. Readers should consult a healthcare professional before making any health-related decisions.
Risks and Concerns
Contaminants
Organ meat supplements can contain a range of contaminants that raise significant safety concerns. Manufacturers sometimes fail to remove active hormones, especially in glandular extracts like adrenal or thyroid supplements. These hormones may disrupt the endocrine system, causing high blood pressure or irregular heart rhythms. Heavy metals such as cadmium, lead, arsenic, chromium, copper, mercury, and nickel often accumulate in animal tissues. Their levels vary by species and region, with some meats showing higher concentrations. Consuming these metals in excess can result in toxicity and long-term organ damage.
Other contaminants include excessive vitamins and minerals, particularly vitamin A and iron. Too much vitamin A can lead to hypervitaminosis A, which causes liver damage, bone loss, and birth defects during pregnancy. Iron overload may result in toxicity, especially for individuals with metabolic disorders. Environmental toxins and pesticides also pose risks, especially when animals are exposed to contaminated feed or water. The lack of standardization and inconsistent testing among brands leads to unpredictable nutrient and contaminant levels, increasing safety concerns for consumers.
Note: The FDA has issued warnings about unlisted hormones in glandular supplements, and the NIH cautions against exceeding safe upper limits of vitamin A due to toxicity risks.
Regulatory Compliance
Regulatory compliance for organ meat supplements varies widely across countries and regions. In the United States, the FDA regulates these products as dietary supplements, requiring no premarket authorization except for new ingredients introduced after 1994. Europe uses a dual approach, classifying products as food ingredients or medicinal substances based on their claims. Australia differentiates between food and therapeutic goods, with complementary medicines often facing less scrutiny unless registered.
Aspect | USA (FDA & DSHEA) | Europe (EMA & EFSA) | Australia (TGA & FSANZ) |
|---|---|---|---|
Product Characterization | Dietary supplement or special food for medical use | Food ingredient or medicinal substance depending on claims | Food or therapeutic good; complementary medicines may require less scrutiny |
Regulation Requirement | No premarketing authorization except for new ingredients | Stricter regulation for medicinal claims | Listed complementary medicines require certification; registered products undergo more scrutiny |
Data Requirements | Clinical and toxicological data for new or high-use ingredients | Biological and toxicology data for medical claims | Certification for listed products; more data for registered products |
Regulatory Authority | FDA monitors safety, labeling, and advertising | EMA for medicines; EFSA for scientific advice and risk assessment | TGA for complementary medicines; FSANZ for food products |
This variability in oversight leads to inconsistent product quality and labeling accuracy. Consumers face safety concerns when brands do not meet strict regulatory standards or fail to provide transparent information.
Excess Intake
Excessive consumption of organ meat supplements introduces several safety concerns. These products are nutrient-dense, but they also contain high cholesterol, which can increase the risk of heart attack or stroke, especially in individuals sensitive to dietary cholesterol. Organ meats have high purine content, which may trigger gout by raising uric acid levels. People with hemochromatosis, a condition involving iron overload, should avoid excess intake to prevent worsening their condition.
Too much vitamin A from supplements can cause toxicity, leading to severe headaches, blurred vision, nausea, dizziness, muscle aches, and coordination problems. In extreme cases, vitamin A toxicity can be fatal. Pregnant or breastfeeding individuals face additional risks, as excess vitamin A may cause birth defects. Experts advise against self-medicating with organ supplements and recommend consulting healthcare providers before use.
Medical Disclaimer: This information is for educational purposes only. Individuals should consult a healthcare professional before using any supplements.
Allergens
Organ meat supplements can introduce allergens that pose safety concerns for sensitive individuals. Manufacturers must address these risks by following strict labeling practices. The most common allergens found in supplements, including organ meat products, align with the “Big Eight” allergens recognized worldwide. These allergens account for the majority of allergic reactions and require clear disclosure on product labels.
Milk
Eggs
Peanuts
Tree nuts
Shellfish
Fish
Soybeans
Wheat
Milk and eggs appear most frequently as allergens in dietary supplements. Some manufacturers use these ingredients as binders, fillers, or part of the capsule composition. Even trace amounts can trigger reactions in people with allergies. In the United States, the Food Allergen Labeling and Consumer Protection Act of 2004 mandates that manufacturers clearly identify these allergens on packaging. Labels often use bold text, different fonts, or background colors to highlight allergen information. A “Contains” statement near the ingredient list helps consumers quickly identify potential risks.
Manufacturers may also include advisory warnings, such as “may contain traces of milk or soy,” although these statements remain voluntary. These warnings alert consumers to possible cross-contamination during production. Regulatory compliance ensures that companies follow these labeling rules, which helps protect consumers from accidental exposure.
Consumers should always read supplement labels carefully. They should look for allergen disclosures and advisory statements before purchasing or consuming organ meat supplements. Individuals with known allergies must remain vigilant, as reactions can range from mild discomfort to severe anaphylaxis. Safety concerns increase when manufacturers fail to provide transparent labeling or when products are processed in facilities that handle multiple allergens.
For those with severe allergies, consulting a healthcare professional before starting any new supplement is essential. Some people may benefit from choosing whole food alternatives, which allow for better control over ingredient exposure. Whole foods also reduce the risk of hidden allergens that sometimes appear in processed supplements.
Medical Disclaimer: The information provided in this article is for informational purposes only and does not constitute medical advice. Readers should consult a healthcare professional before making any health-related decisions.
Making Informed Choices
Key Markers
Shoppers can identify high-quality organ meat supplements by focusing on several key markers. Trusted brands display certifications such as GMP, FDA registration, and ISO 17025 lab accreditation. These markers show that the manufacturer follows strict safety and quality standards. Clear sourcing information, like grass-fed or pasture-raised claims, signals ethical practices. Brands that offer detailed Certificates of Analysis (COA) for each batch demonstrate transparency. Products with short ingredient lists and no fillers or artificial additives also stand out as nutrient-dense superfoods.
Steps Before Buying
Consumers should take specific steps before purchasing supplements.
Research the Brand: Check the company’s reputation and read independent reviews.
Verify Certifications: Look for GMP, FDA, ISO, and organic certifications on the label or website.
Request a COA: Ask for a recent COA from an ISO 17025-accredited lab.
Examine the Ingredient List: Choose products with only freeze-dried organs and no unnecessary fillers.
Check Sourcing: Prefer supplements made from grass-fed, pasture-raised cattle.
Contact Customer Service: Reach out with questions about sourcing, testing, or allergens.
Review Allergen Statements: Ensure the product does not contain ingredients that may cause allergic reactions.
Tip: Taking these steps helps consumers avoid low-quality products and ensures they select safe, effective supplements.
Red Flags
Certain warning signs suggest a supplement may not meet safety or quality standards.
The brand refuses to provide a COA or hides testing results.
The ingredient list includes fillers, artificial colors, or preservatives.
The product lacks clear sourcing information or uses vague terms like “proprietary blend.”
The company has poor customer reviews or unresolved complaints.
The label does not mention third-party testing or certifications.
Red Flag | What It Means |
|---|---|
No COA available | Lack of transparency |
Fillers or additives | Lower purity and possible allergens |
Vague sourcing | Unknown quality or origin |
No certifications listed | Unverified safety or quality |
Note: Consumers should avoid supplements with these red flags to protect their health and investment.
Medical Disclaimer: The information provided in this article is for informational purposes only and does not constitute medical advice. Readers should consult a healthcare professional before making any health-related decisions.
Strict standards in organ meat supplements protect consumer health and ensure product quality. Shoppers should always check for certifications, third-party testing, and transparent labeling. High-quality supplements offer concentrated nutrients, but whole foods provide balanced nutrition and fewer risks.
Note: This article provides general information only. It does not replace medical advice. Readers should consult a healthcare professional before using any supplement.
FAQ
What are organ meat supplements made from?
Organ meat supplements contain freeze-dried organs, such as liver, heart, kidney, or spleen, from grass-fed or pasture-raised cattle. Manufacturers process these organs into powder and encapsulate them for easy consumption.
How can consumers verify supplement quality?
Consumers should check for GMP, FDA, and ISO certifications. They can request a Certificate of Analysis (COA) from the manufacturer. Reputable brands provide third-party lab results and clear sourcing information.
Are organ meat supplements safe for everyone?
Most healthy adults tolerate organ meat supplements. Individuals with allergies, iron overload, or certain medical conditions should consult a healthcare provider before use. Pregnant or breastfeeding women need professional guidance.
Do organ meat supplements contain allergens?
Some supplements may contain allergens, such as milk or eggs, used as binders or fillers. Consumers should always read labels and check for allergen statements before purchasing.
How do organ meat supplements compare to eating whole organs?
Supplements offer convenience and concentrated nutrients. Whole organs provide fiber and a broader nutrient profile. Both options support health, but whole foods may reduce the risk of excessive nutrient intake.
Can athletes use organ meat supplements?
Many brands test supplements for banned substances. Athletes should choose products with third-party certifications, such as NSF Certified for Sport or Informed Choice, to ensure safety and compliance.
What should consumers avoid when choosing a supplement?
Consumers should avoid products without COAs, unclear sourcing, or those containing fillers and artificial additives. Negative reviews and lack of certifications also signal poor quality.
Medical Disclaimer: This information is for educational purposes only. It does not constitute medical advice. Readers should consult a healthcare professional before making any health-related decisions.


